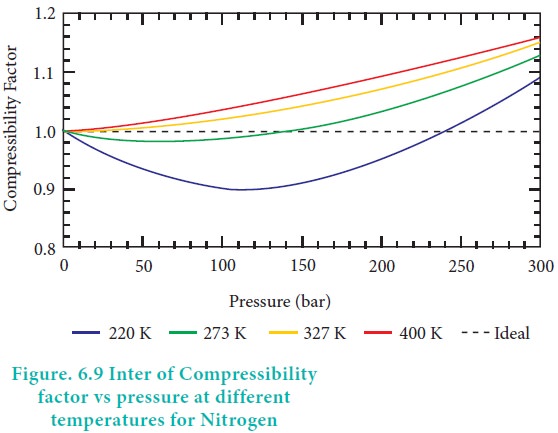
The given graph represent the variations of Z Compressibility

The compressibility factor is actually a factor that corrects the actual value of the gas versus the ideal gas. Let us learn and understand this concept.
What is the compressibility factor, and how does it vary with an increase in temperature and pressure? Watch this video to get the answer. This is an importa

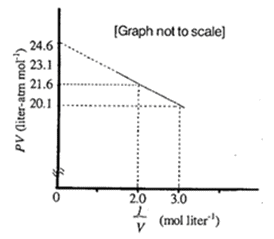
For different ideal gases d/P versus P variations at definite
03-01-2018) Mole Concept, Gaseous and Surface Chemistry Free MCQ
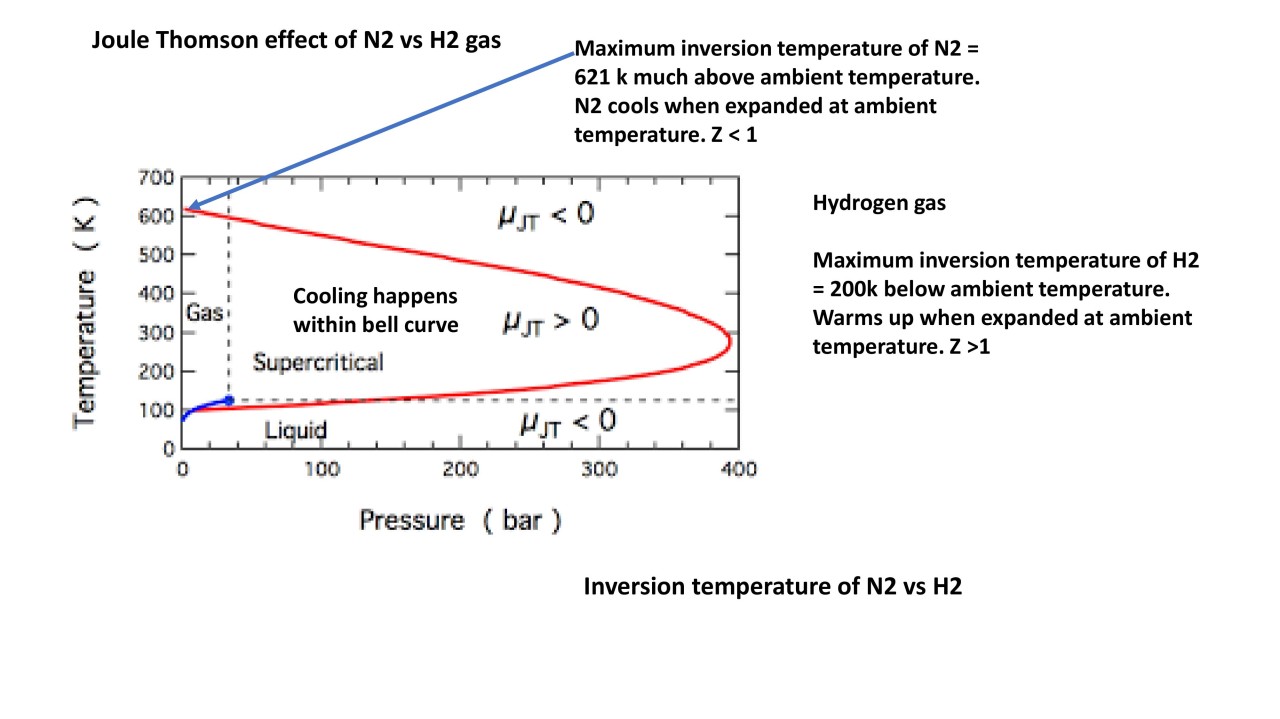
Joule Thomson effect [JT]: A short review
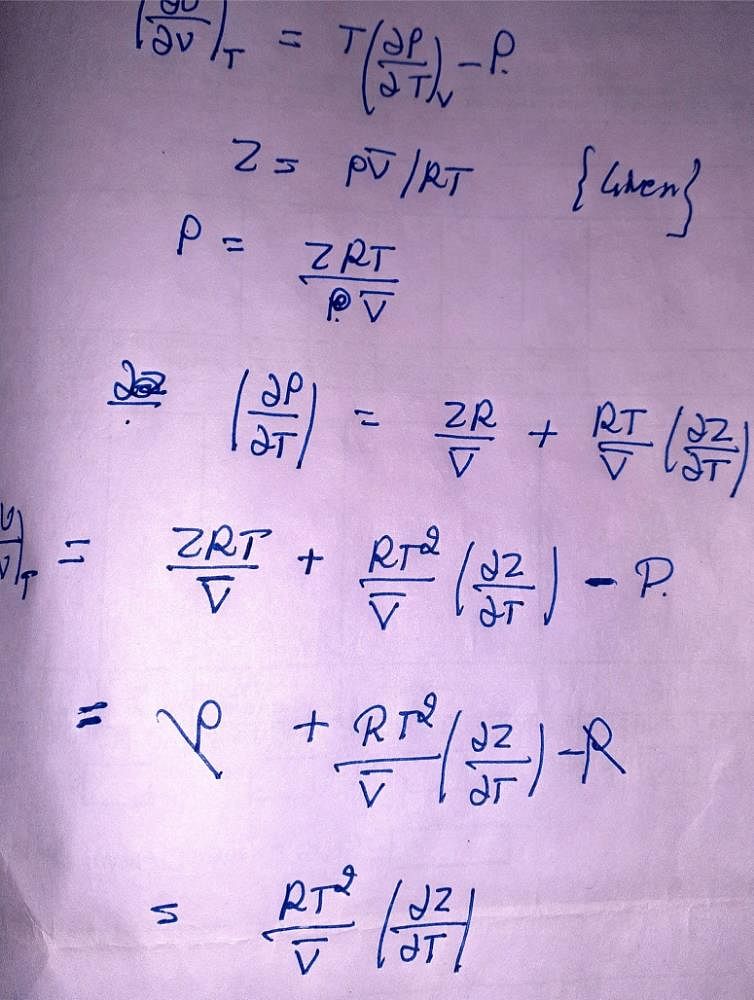
The internal pressureof a real gas is related to the
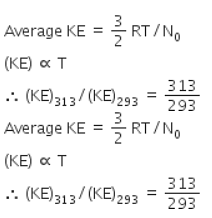
Gaseous State Questions for JEE exam - Free Online All questions
Prandtl number effects on the hydrodynamic stability of compressible boundary layers: flow–thermodynamics interactions, Journal of Fluid Mechanics

Entropic barrier of water permeation through single-file channels

Gas C is a real gas and we can find 'a' and 'b' if intersection data i

For the different ideal gases Bigg(frac{d}{P}Bigg) versus P
The given graph represents the variations of compressibility factor Z=PV/..

Compressibility factor - Wikipedia
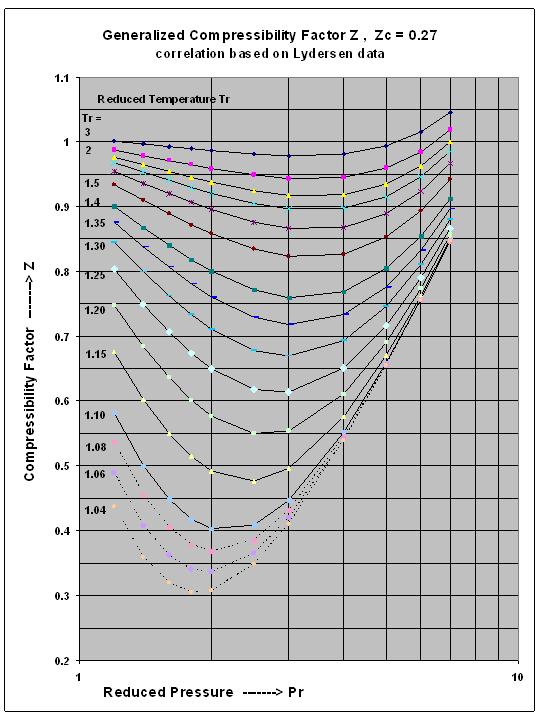
Super-critical Fluid Compressibility Factor Z , for Intermediate Reduced Pressure Range, a new correlation for excel spreadsheets

The given graph represents the variation of compressibility factor